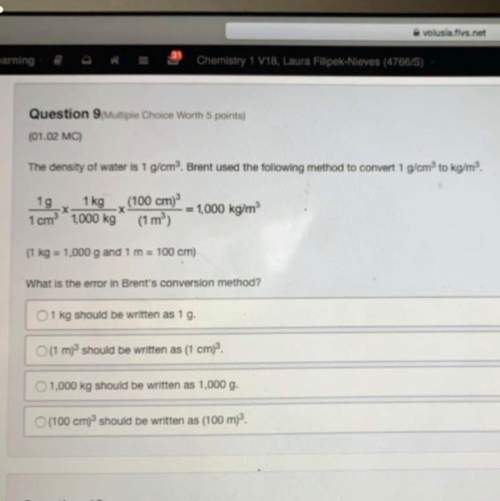
Chemistry, 25.11.2021 15:30 mayachojnicki
Help Help Help Help Help Help Help Help Help Help Help Help Help Help Help Help Help Help Help Help Help Help
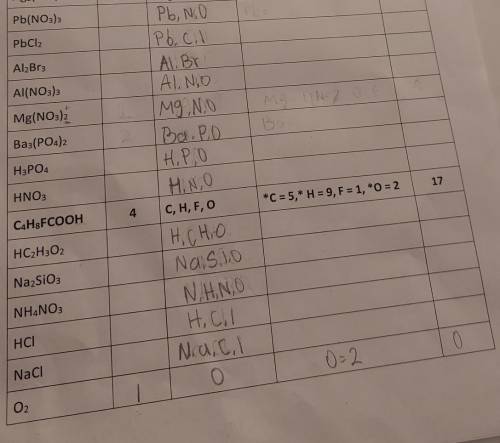

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 20:00, 20calzoy
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3


Chemistry, 23.06.2019 05:00, kayranicole1
How is electrolysis most commonly used to produce an energy source? a - splitting water molecules produces oxygen, which organisms breathe to fuel their bodies. b - splitting water molecules produces hydrogen gas, which is used to power machines through hydrogen fuel cells. c - splitting carbon dioxide molecules produces coal, a form of carbon that can be burned to produce heat. d - splitting carbon dioxide molecules produces natural gas, which can be burned to generate electricity in power plants.
Answers: 1
You know the right answer?
Help Help Help Help Help Help Help Help Help Help Help Help Help Help Help Help Help Help Help Help...
Questions in other subjects:

Mathematics, 11.09.2020 23:01

Mathematics, 11.09.2020 23:01

Social Studies, 11.09.2020 23:01

Spanish, 11.09.2020 23:01

Mathematics, 11.09.2020 23:01

Mathematics, 11.09.2020 23:01

Mathematics, 11.09.2020 23:01

Mathematics, 11.09.2020 23:01

Physics, 11.09.2020 23:01

Mathematics, 11.09.2020 23:01