
Chemistry, 08.11.2021 14:00 jjmage2206
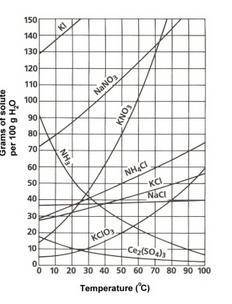
A sample of potassium nitrate (49.0 g) is dissolved in 100 g of water at 100oC. This solution is _.

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 08:30, Blaise2653
Joan writes four numbers on the board in standard form, and then she writes their scientific notation
Answers: 1

Chemistry, 22.06.2019 10:00, Cythina2007
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1
You know the right answer?
A sample of potassium nitrate (49.0 g) is dissolved in 100 g of water at 100oC. This solution is _....
Questions in other subjects:



History, 22.02.2021 21:50


English, 22.02.2021 21:50

English, 22.02.2021 21:50

Mathematics, 22.02.2021 21:50






