
Chemistry, 02.10.2021 17:30 bagofmud8339
Please help! It is due in an hour and I'm solving other problems in this assignment, but just can't get this one.
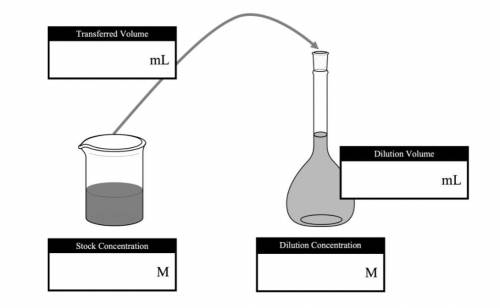
If you use a volumetric pipet to transfer 10 mL of a 0.401 M stock solution into a 50 mL volumetric flask and then filled the flask to the calibration mark with deionized water, what would the concentration (in M) of the new solution be? Filling in a dilution diagram like the one below, may help you with this calculation.
Edit: 0.0802 M
Explanation: C1V1 = C2V2
We need to solve for C2 so set the formula to fit that.
C2 = C1V1/V2
C2 = (10 mL • 0.401 M)/ 50 mL = 0.0802M

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:30, greekfreekisdbz
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 3


Chemistry, 22.06.2019 12:20, jessicasbss6840
Adeuteron, 21h, is the nucleus of a hydrogen isotope and consists of one proton and one neutron. the plasma of deuterons in a nuclear fusion reactor must be heated to about 3.02×108 k . what is the rms speed of the deuterons? express your answer using two significant figures.
Answers: 1

You know the right answer?
Please help! It is due in an hour and I'm solving other problems in this assignment, but just can't...
Questions in other subjects:


Mathematics, 18.03.2021 04:20

Health, 18.03.2021 04:20

Mathematics, 18.03.2021 04:20



Mathematics, 18.03.2021 04:20

Health, 18.03.2021 04:20

Mathematics, 18.03.2021 04:20

Mathematics, 18.03.2021 04:20



