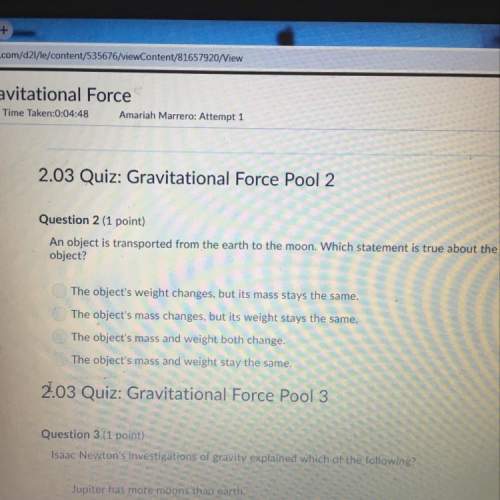
Please Help
11. How many moles of gas does it take to occupy 120 liters at a pressure of 2.3 atmospheres and a temperature of 340 K?
12. If I have a 50-liter container that holds 45 moles of gas at a temperature of 2000 C, what is the pressure inside the container?
13. It is not safe to put aerosol canisters in a campfire, because the pressure inside the canisters gets very high and they can explode. If I have a 1.0-liter canister that holds 2 moles of gas, and the campfire temperature is 14000 C, what is the pressure inside the canister?
14. How many moles of gas are in a 30-liter scuba canister if the temperature of the canister is 300 K and the pressure is 200 atmospheres?
15. I have a balloon that can hold 100 liters of air. If I blow up this balloon with 3 moles of oxygen gas at a pressure of 1 atmosphere, what is the temperature of the balloon?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:50, chem1014
Given the following information: mass of proton = 1.00728 amu mass of neutron = 1.00866 amu mass of electron = 5.486 × 10^-4 amu speed of light = 2.9979 × 10^8 m/s calculate the nuclear binding energy (absolute value) of 3li^6. which has an atomic mass of 6.015126 amu. j/mol.
Answers: 2

Chemistry, 23.06.2019 00:20, jessicamcummins
What type of context clue you understand the meaning of quandary?
Answers: 3

Chemistry, 23.06.2019 10:00, anonymous176
An uncovered pot of water lies out in the sun. which statements correctly describe what happens at the surface of the liquid water? 1. the vapor pressure remains constant regardless of the water temperature. 2. the vapor pressure is produced by water molecules that have evaporated. 3. the vapor pressure increases as the sun heats the water in the pot. 4. evaporation stops once the vapor pressure reaches a certain point. 5. evaporation and condensation both occur on the liquid’s surface.
Answers: 3
You know the right answer?
Please Help
11. How many moles of gas does it take to occupy 120 liters at a pressure of 2.3 atmos...
Questions in other subjects:


History, 29.01.2021 22:50






History, 29.01.2021 22:50

Mathematics, 29.01.2021 22:50