
Chemistry, 04.09.2021 16:30 mairealexander87
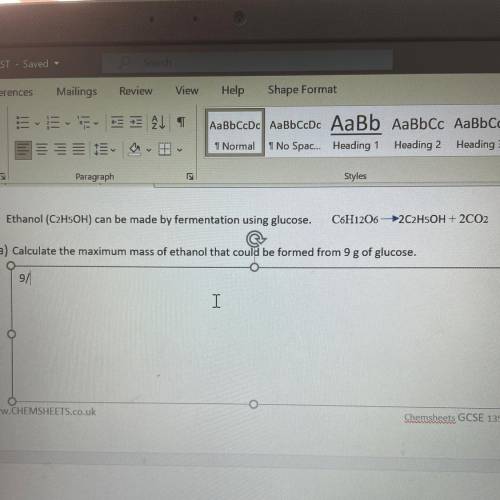
Ethanol (C2H5OH)can be made by fermentation using glucose. C6H12O6 –>2C2H5OH+2CO2 Calculate the maximum mass of ethanol that could be formed from 9g of glucose.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:00, lydiadmanautou04
Write a hypothesis that answers the lesson question, “while observing a chemical reaction, how can you tell which reactant is limiting? ” hypothesis: if a substance is the limiting reactant, then . . because . .
Answers: 1

Chemistry, 22.06.2019 08:30, MacenParisi
In the reaction between a crushed antacid tablet and vinegar what gas is emitted
Answers: 2

Chemistry, 22.06.2019 19:00, innocentman69
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated complex stage, allowing products to form immediately. b. the catalyst lowers the energy level of the reactants, making it easier for them to react. c. the catalyst makes it easier for the activated complex to form, lowering the activation energy. d. the catalyst raises the energy level of the products, making the reaction finish sooner. reset next
Answers: 1

Chemistry, 23.06.2019 09:00, floressavanna15
What properties would have caused early researchers to name hydrogen "inflammable air”
Answers: 3
You know the right answer?
Ethanol (C2H5OH)can be made by fermentation using glucose. C6H12O6 –>2C2H5OH+2CO2
Calculate the...
Questions in other subjects:


Mathematics, 07.11.2019 16:31


English, 07.11.2019 16:31



Geography, 07.11.2019 16:31

History, 07.11.2019 16:31




