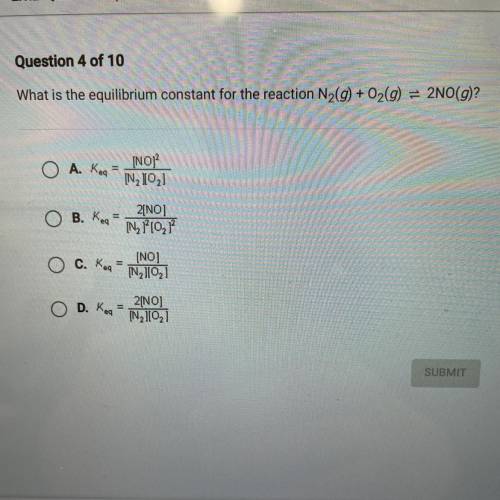
What is the equilibrium constant for the reaction N2(g) + O2(g) = 2NO(g)?
...


Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:50, jonmorton159
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3


Chemistry, 22.06.2019 17:40, adantrujillo1234
Areaction in which products can react to re-form reactants is
Answers: 1

Chemistry, 23.06.2019 04:31, 24swimdylanoh
What are the coefficients that will balance the skeleton equation below? n2 + h2 → nh3
Answers: 1
You know the right answer?
Questions in other subjects:

Mathematics, 20.05.2021 03:40

Mathematics, 20.05.2021 03:40

English, 20.05.2021 03:40

Social Studies, 20.05.2021 03:40


Mathematics, 20.05.2021 03:40

History, 20.05.2021 03:40