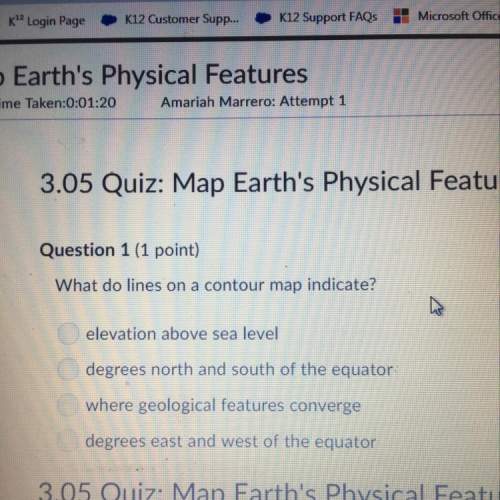
Chemistry, 24.08.2021 16:20 queentynaisa
The bromination of acetone is acid-catalyzed.
CH3COCH3 + Br2 CH3COCH2Br + H+ + Br -
The rate of disappearance of bromine was measured for several different concentrations of acetone, bromine, and H+ ions at a certain temperature.
[CH3COCH3] [Br2] [H+] Rate of disappearance of Br2 (M/s)
0.30 0.050 0.050 5.7 10-5
0.30 0.10 0.050 5.7 10-5
0.30 0.050 0.10 1.2 10-4
0.40 0.050 0.20 3.1 10-4
0.40 0.050 0.050 7.6 10-5
(a) What is the rate law for the reaction?
Rate = k[CH3COCH3][H+][Br2]
Rate = k[Br2][H+]
Rate = k[CH3COCH3][H+]
Rate = k[CH3COCH3][Br2]
(b) Determine the rate constant.

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 00:00, 20jessicacabriales
The pressure in a fluid is affected by which characteristics of that fluid
Answers: 1

Chemistry, 22.06.2019 06:00, citlalli30
An atom of sodium-23 (atomic number = 11) has a positive charge of +1. give this information, how many electrons does it have? how many proteins and neutrons does this atom have
Answers: 2

Chemistry, 22.06.2019 08:30, waterborn7152
Which common material is an example of a polymer? (25 pts) a. steel b. plastic c. petroleum d. rubbing alcohol
Answers: 2
You know the right answer?
The bromination of acetone is acid-catalyzed.
CH3COCH3 + Br2 CH3COCH2Br + H+ + Br -
The rate...
The rate...
Questions in other subjects:

Mathematics, 05.12.2019 21:31

Physics, 05.12.2019 21:31



Mathematics, 05.12.2019 21:31

History, 05.12.2019 21:31



Biology, 05.12.2019 21:31