
Chemistry, 23.08.2021 21:00 brookealexis5768
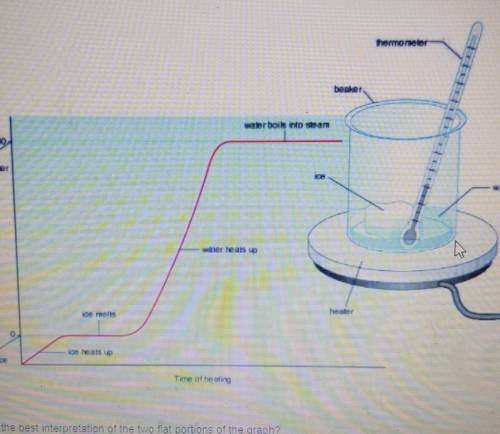
Which is the best interpretation of the two flat portions of the graph
A. These show where kinetic energy is increased
B. These show where temperature is at 0 Celsius
C. These show where thermal energy does not exist
D. These show were changes of state are occurring

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 06:30, dpchill5232
Suppose a lab group reports a ppercent yield of sand of 105. is it really possible to collect more sand than was originally represented? what is the possible explanation for the extra product?
Answers: 2

Chemistry, 22.06.2019 14:30, clemsongirl5392
Select all that apply. using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 (s) pb+2(aq) + 2cl -(aq). the concentration of the products yield a ksp of 2.1 x 10-2:
Answers: 2

Chemistry, 22.06.2019 18:00, kyllow5644
Answer asap need to be answered by wednesday morning explain how a buffer works, using an ethanoic acid / sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 3
You know the right answer?
Which is the best interpretation of the two flat portions of the graph
A. These show where kinetic...
Questions in other subjects:

Mathematics, 31.08.2019 01:30

Social Studies, 31.08.2019 01:30


Mathematics, 31.08.2019 01:30

Physics, 31.08.2019 01:30

Computers and Technology, 31.08.2019 01:30




Mathematics, 31.08.2019 01:30



