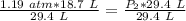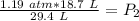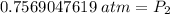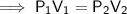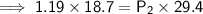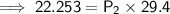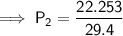Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:30, deidaraXneji
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 3


Chemistry, 23.06.2019 04:20, milkshakegrande101
The equation below shows the reaction of zinc with hydrochloric acid (hcl). zn (s) + 2 hcl (aq) —> zncl2 (aq) + h2 (g) what will happen if the concentration of hcl is decreased? a. more zncl2 will be produced. b. the reaction rate will slow down. c. the hydrochloric acid will become more acidic. d. the reaction will produce water instead of hydrogen gas.
Answers: 1

Chemistry, 23.06.2019 10:30, piratesfc02
When a wire with a current is placed in a magnetic field, electrical energy is transformed into mechanical energy select the best answer from the choices provided t f
Answers: 2
You know the right answer?
A sample of oxygen gas at a pressure of 1.19 atm and a temperature of 24.4 °C, occupies a volume of...
Questions in other subjects:


English, 25.04.2020 02:19



History, 25.04.2020 02:21



Mathematics, 25.04.2020 02:23

English, 25.04.2020 02:28


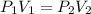
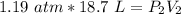

 . It is being multiplied by 29.4 liters. The inverse operation of multiplication is division. Divide both sides of the equation by 29.4 L.
. It is being multiplied by 29.4 liters. The inverse operation of multiplication is division. Divide both sides of the equation by 29.4 L.