
It is often possible to change a hydrate into an anhydrous compound by heating it to drive off the water (dehydration). A 43.19 gram sample of a hydrate of MgBr2 was heated thoroughly in a porcelain crucible, until its weight remained constant. After heating, 27.21 grams of the anhydrous compound remained. What is the formula of the hydrate?

Answers: 2


Other questions on the subject: Chemistry




Chemistry, 22.06.2019 16:00, matt16913
Which of the following is the correct definition of chemical energy? a. energy an object has because of its motion or position b. energy resulting from the flow of charged particles, such as electrons or ions c. energy produced from the splitting of atoms d. energy stored in chemical bonds of molecules
Answers: 1
You know the right answer?
It is often possible to change a hydrate into an anhydrous compound by heating it to drive off the w...
Questions in other subjects:


Mathematics, 24.04.2020 20:15

Business, 24.04.2020 20:15


Mathematics, 24.04.2020 20:15

Biology, 24.04.2020 20:15




 .
. denote the number of
denote the number of 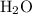 formula units for every
formula units for every  formula unit. The formula of the hydrate would be
formula unit. The formula of the hydrate would be  .
. :
: 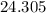 .
. :
: 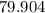 .
. :
:  .
. :
: 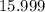 .
. .
. .
.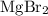 . There was
. There was 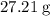 of this compound. Calculate the number of moles of formula units in that much of this compound:
of this compound. Calculate the number of moles of formula units in that much of this compound: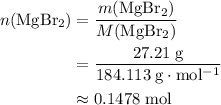 .
.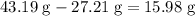 of water
of water 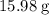 of
of 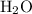 :
: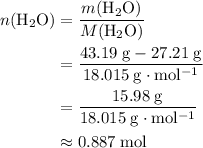 .
. of
of 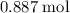 of
of 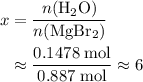 .
.

