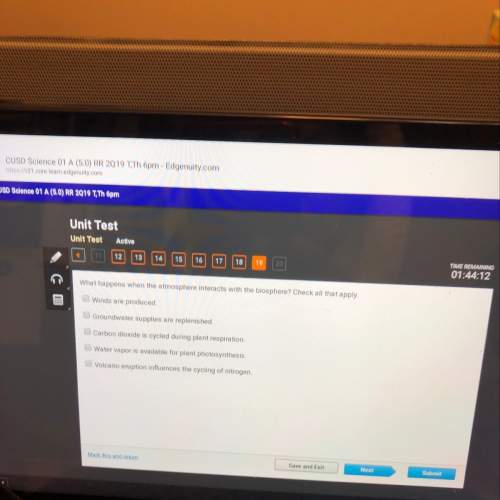
Chemistry, 05.08.2021 04:10 justhereforanswers13
A student reacts benzene, C6H6, with bromine, Br2, to prepare bromobenzene, C6H5Br, and HBr. C6H6 Br2 --> C6H5Br HBr What is the theoretical yield of bromobenzene in this reaction when 0.31 moles of benzene reacts with 0.46 moles of bromine

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, kingteron5870
Ionic compounds are made of ions, and yet the overall charge of an ionic compound is neutral. why?
Answers: 1

Chemistry, 22.06.2019 12:30, hala201490
Place the elements below in order of decreasing ionization energy. aluminum(al) chlorine(cl) magnesium (mg) sulfur(s)
Answers: 1

Chemistry, 22.06.2019 12:30, ethanw8973
If 22.5 liters of oxygen reacted with excess of hydrogen, how many liters of water vapor could be produced?
Answers: 3
You know the right answer?
A student reacts benzene, C6H6, with bromine, Br2, to prepare bromobenzene, C6H5Br, and HBr. C6H6 Br...
Questions in other subjects:

Mathematics, 07.11.2019 22:31


Mathematics, 07.11.2019 22:31



Spanish, 07.11.2019 22:31

SAT, 07.11.2019 22:31

Mathematics, 07.11.2019 22:31