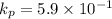Chemistry, 28.07.2021 04:30 markcardenas2756
Hydrogen chloride decomposes to form hydrogen and chlorine, like this:
2HCl(g) + H2(g) â Cl2(g)
Also, a chemist finds that at a certain temperature the equilibrium mixture of hydrogen chloride, hydrogen, and chlorine has the following composition:
compound pressure at equilibrium
HCl 84.4 atm
H2 77.9 atm
Cl2 54.4
Required:
Calculate the value of the equilibrium constant for this reaction. Round your answer to significant digits.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:00, bakoeboo
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2

Chemistry, 22.06.2019 10:30, shaylawaldo11
Apiece of metal with a length of 1.42 cm was measured using four different instruments. which of the following measurements is the most accurate?
Answers: 3

Chemistry, 22.06.2019 14:30, Playboycxm
Chemistry worksheet - i am not sure what they are asking for exactly?
Answers: 1

You know the right answer?
Hydrogen chloride decomposes to form hydrogen and chlorine, like this:
2HCl(g) + H2(g) â Cl2(g)
Questions in other subjects:


Chemistry, 03.07.2019 23:30




Mathematics, 03.07.2019 23:30

Biology, 03.07.2019 23:30


Chemistry, 03.07.2019 23:30

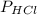 = 84.4 atm
= 84.4 atm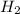 ,
, 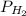 = 77.9 atm
= 77.9 atm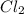 ,
, 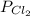 = 54.4 atm
= 54.4 atm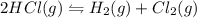
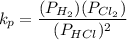
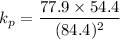
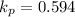
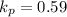 (in 2 significant figures)
(in 2 significant figures)