
Chemistry, 28.07.2021 03:50 lizzyhearts
Urgent need help
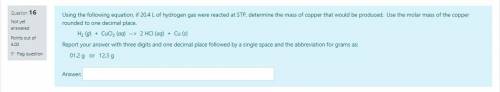
I am doing a test which is due today, so please

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 15:30, lovebaeforlife351
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins. co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1

Chemistry, 22.06.2019 18:10, bri9263
Consider the following reaction at equilibrium: c(s)+h2o(g)⇌co(g)+h2(g) predict whether the reaction will shift left, shift right, or remain unchanged upon each of the following disturbances. a) c is added to the reaction mixture. b) h2ois condensed and removed from the reaction mixture c) co is added to the reaction mixture d) h2 is removed from the reaction mixture.
Answers: 3

Chemistry, 22.06.2019 21:30, Turtlelover05
How can the periodic table be used to predict the behavior of elements?
Answers: 1
You know the right answer?
Urgent need help
I am doing a test which is due today, so please
...
I am doing a test which is due today, so please
...
Questions in other subjects:


Chemistry, 28.12.2019 06:31

Mathematics, 28.12.2019 06:31

Biology, 28.12.2019 06:31

History, 28.12.2019 06:31







