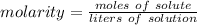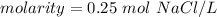Chemistry, 28.07.2021 02:10 tanviknawale
What is the molarity of a solution that contains 0.75 mol Naci in 3.0 L of solution? Select one: O a. 4.0 M O b. 2.3 M O d. 3.8 M O d. 0.25 M Clear my choice

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:30, caitybugking
Type the correct answer in the box. spell all words correctly. what is the correct term for living the most sustainable life you can within your current circumstances? when your are being as sustainable as you can within your current lifestyle, you are said to be sustainability.
Answers: 3

Chemistry, 22.06.2019 08:00, PrincessKeliah5538
Me i dont know what to do! the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 12:00, vannitling12p4w44f
What is the percentage of hydrogen in nitrogen trihydride
Answers: 1

You know the right answer?
What is the molarity of a solution that contains 0.75 mol Naci in 3.0 L of solution? Select one: O a...
Questions in other subjects:

Mathematics, 25.06.2019 13:30

Mathematics, 25.06.2019 13:30


Mathematics, 25.06.2019 13:30

Biology, 25.06.2019 13:30

Mathematics, 25.06.2019 13:30

Mathematics, 25.06.2019 13:30