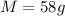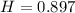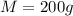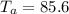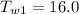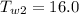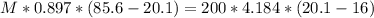Chemistry, 27.07.2021 01:00 queenkimm26
A sample of aluminum, which has a specific heat capacity of , is put into a calorimeter (see sketch at right) that contains of water. The temperature of the water starts off at . When the temperature of the water stops changing it's . The pressure remains constant at .Calculate the initial temperature of the aluminum sample. Be sure your answer is rounded to significant digits.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 23.06.2019 07:50, alexusnicole817
Asolution is produced in which water is the solvent and there are four solutes. which of the solutes can dissolve better if the solution is heated?
Answers: 1


Chemistry, 23.06.2019 15:00, ericperkins10ox0b27
In two or more complete sentences describe all of the van der waals forces that exist between molecules of sulfur dioxide, so2.
Answers: 1

Chemistry, 23.06.2019 19:30, FantasticFerret
What is the pressure of 5.0 mol nitrogen gas in a 2.0 l container at 268 k (the universal gas constant is 0.0821 l•atm/mol•k) a. 55 atm b. 8.8 atm c. 0.018 atm d. 220 atm
Answers: 1
You know the right answer?
A sample of aluminum, which has a specific heat capacity of , is put into a calorimeter (see sketch...
Questions in other subjects:

Physics, 18.12.2020 20:50




History, 18.12.2020 20:50


Biology, 18.12.2020 20:50


Mathematics, 18.12.2020 20:50

Social Studies, 18.12.2020 20:50