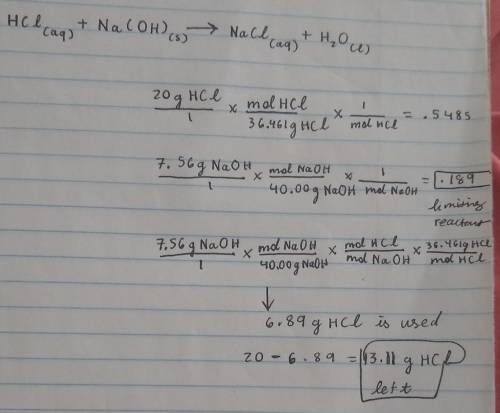Chemistry, 23.07.2021 03:50 morrowdrew
Aqueous hydrochloric acid HCl will react with solid sodium hydroxide NaOH to produce aqueous sodium chloride NaCl and liquid water H2O. Suppose 20. g of hydrochloric acid is mixed with 7.56 g of sodium hydroxide. Calculate the minimum mass of hydrochloric acid that could be left over by the chemical reaction. Round your answer to 2 significant digits.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:30, alexisfaithsmith
How many atoms are in 1.4 mil of phosphorus trifluoride (pf3)
Answers: 3

Chemistry, 23.06.2019 00:30, DragonLovely
•hydration •dissociation •dissolving which one goes to which
Answers: 1

Chemistry, 23.06.2019 01:00, daniel1480
Which fossil fuel is mainly used for heating and cooking? a. electricity b. coal c. petroleum d. natural gas
Answers: 2
You know the right answer?
Aqueous hydrochloric acid HCl will react with solid sodium hydroxide NaOH to produce aqueous sodium...
Questions in other subjects:


English, 27.06.2019 12:40


Mathematics, 27.06.2019 12:40




History, 27.06.2019 12:40