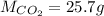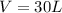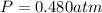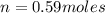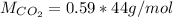Carbon dioxide gas is collected at 27.0 oC in an evacuated flask with a measured volume of 30.0L. When all the gas has been collected, the pressure in the flask is measured to be 0.480atm. Calculate the mass and number of moles of carbon dioxide gas that were collected.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:10, lilyjordan5972
How do forces between particles in gases compare to forces in the other states of matter? o a. the forces in gases are stronger than forces in solids but weaker than forces in liquids. o b. the forces in gases are weaker than forces in solids but stronger than forces in liquids. o c. the forces in gases are weaker than forces in solids and liquids. o d. the forces in gases are stronger than forces in solids and liquids. submit
Answers: 1

Chemistry, 21.06.2019 22:30, mauifrifer3986
Omg imgonnafailnfiedkla use complete sentences to explain how the mass of hydrogen is conserved during cellular respiration.
Answers: 1

You know the right answer?
Carbon dioxide gas is collected at 27.0 oC in an evacuated flask with a measured volume of 30.0L. Wh...
Questions in other subjects:

Mathematics, 04.05.2021 20:50



English, 04.05.2021 20:50




Mathematics, 04.05.2021 20:50

English, 04.05.2021 20:50

Computers and Technology, 04.05.2021 20:50