

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 12:00, Unknowndragon42
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2


You know the right answer?
A 0.4272 g sample of an element contains 2.241 x 10 ^21 atoms . what is the symbol of the element?...
Questions in other subjects:


Mathematics, 31.07.2019 05:30







Mathematics, 31.07.2019 05:30

Mathematics, 31.07.2019 05:30

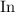 (indium.)
(indium.)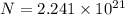 .
. , the number of atoms by the Avogadro constant,
, the number of atoms by the Avogadro constant, 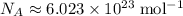 , would give the number of moles of atoms in this sample:
, would give the number of moles of atoms in this sample: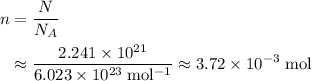 .
. . Estimate the average mass of one mole of atoms in this sample:
. Estimate the average mass of one mole of atoms in this sample: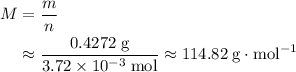 .
. in this example) is numerically equal to the average atomic mass of that element. Refer to a modern periodic table and look for the element with average atomic mass
in this example) is numerically equal to the average atomic mass of that element. Refer to a modern periodic table and look for the element with average atomic mass 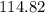 . Indium,
. Indium, 

