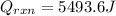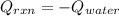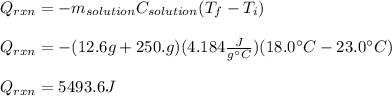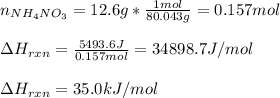Chemistry, 19.07.2021 15:30 kevin72937
A student dissolves 12.6g of amonium nitrate(NH4NO3) in 250.g of water in a well-insulated open cup. She then observed the temperature of the water fall from 23.0°C to 18°C over the course of 6.1 minutes.
NH4NO3 â NH4+ (aq) + NO3^-(aq)
a. Is this reaction exothermic, endothermic, or neither?
b. If you said the reaction was exothermic or calculate the amount of heat that was released or absorbed by the reaction in this case.
c. Calculate the reaction enthalpy ÎHrxn per mole of NH4NO3.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, applejulianamoreno
If a polyatomic ionic compound has gained two hydrogen ions, then how does its name begin?
Answers: 3

Chemistry, 22.06.2019 06:00, citlalli30
An atom of sodium-23 (atomic number = 11) has a positive charge of +1. give this information, how many electrons does it have? how many proteins and neutrons does this atom have
Answers: 2

Chemistry, 22.06.2019 07:20, letsbestupidcx2314
Which of these conditions most likely produces an unstable isotope?
Answers: 1

Chemistry, 22.06.2019 14:00, ashlynneboogs0056
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3
You know the right answer?
A student dissolves 12.6g of amonium nitrate(NH4NO3) in 250.g of water in a well-insulated open cup....
Questions in other subjects:





Chemistry, 24.09.2021 03:30

Mathematics, 24.09.2021 03:30

Mathematics, 24.09.2021 03:30


Mathematics, 24.09.2021 03:30