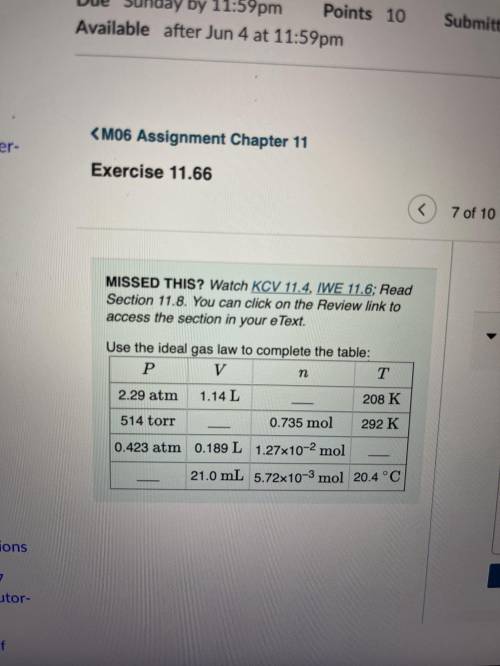
Complete the table.
Need it ASAP.
...

Chemistry, 17.07.2021 19:30 offensiveneedle
Complete the table.
Need it ASAP.

Answers: 2


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 14:50, jonmorton159
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3

Chemistry, 22.06.2019 19:00, innocentman69
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated complex stage, allowing products to form immediately. b. the catalyst lowers the energy level of the reactants, making it easier for them to react. c. the catalyst makes it easier for the activated complex to form, lowering the activation energy. d. the catalyst raises the energy level of the products, making the reaction finish sooner. reset next
Answers: 1
You know the right answer?
Questions in other subjects:




Chemistry, 17.02.2021 22:00

Advanced Placement (AP), 17.02.2021 22:00


Mathematics, 17.02.2021 22:00


Computers and Technology, 17.02.2021 22:00




