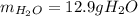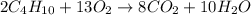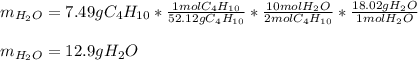Chemistry, 17.07.2021 05:40 morgannwaldrupp
Calculate the mass of water produced when 7.49 g of butane reacts with excess oxygen.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:00, brapmaster764
What is the formula that this ionic compounds could form sr2+p3-o2-
Answers: 3

Chemistry, 22.06.2019 12:00, 1963038660
What are the first two quantum numbers for the electrons located in subshell 4d? what are the first three quantum numbers for the electrons located in subshell 2s? how many electrons can be held in a sublevel l = 3? how many electrons can be held in the energy level n = 4? how many electrons in an atom can share the quantum numbers n = 4 and l = 3?
Answers: 1

Chemistry, 22.06.2019 12:30, UaRemomGAY
If anyone would be able to me out with these three questions it would be these are from the chem 2202 course.
Answers: 3

Chemistry, 22.06.2019 16:50, brandiwingard
What is conserved in the reaction shown below? h2(g) + cl2 (g) --> 2hcl(g)a. mass onlyb. mass and moles onlyc. mass, moles, and molecules onlyd. mass, moles, molecules, and volume
Answers: 2
You know the right answer?
Calculate the mass of water produced when 7.49 g of butane reacts with excess oxygen....
Questions in other subjects:

History, 17.08.2019 02:10

Mathematics, 17.08.2019 02:10



Mathematics, 17.08.2019 02:10

History, 17.08.2019 02:10


Mathematics, 17.08.2019 02:10


Mathematics, 17.08.2019 02:10