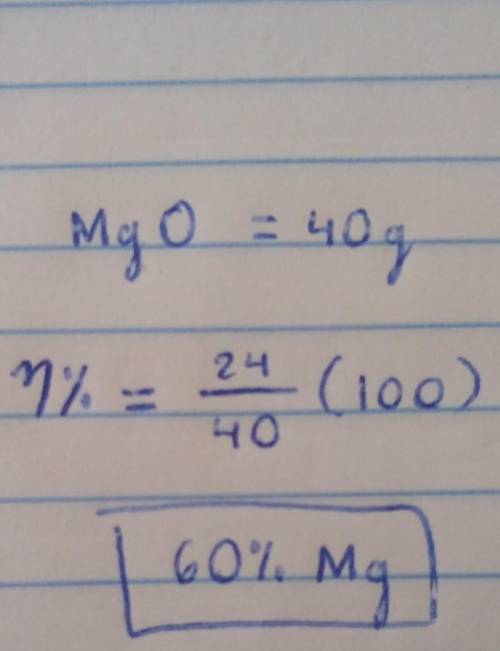Chemistry, 17.07.2021 01:00 rachelreed
1a. calculate the relative formula mass of magnesium oxide when the relative atomic masses are O=16 Mg=24 1b. calculate the percentage by mass of magnesium in magnesium oxide

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 20:00, kalcius9698
What is the molarity of the solution produced when 145 g of nacl is dissolved in sufficient water to prepare 2.75 l of solution?
Answers: 1

Chemistry, 23.06.2019 14:20, BaileyElizabethRay
What kind of chemical reaction does the chemical equation sodium + chlorine → sodium chloride represent? a. combustion b. decomposition c. single replacement d. synthesis
Answers: 1

Chemistry, 23.06.2019 14:30, jenorajordan5387
An atom of element x has one more shell of electrons than an atom of beryllium, but it has one less valance electron than beryllium. which element is x
Answers: 1

Chemistry, 23.06.2019 16:00, naiomireyes74p2aybs
Be sure to answer all parts. the catalytic destruction of ozone occurs via a two-step mechanism, where x can be any of several species: (1) x + o3 → xo + o2 [slow] (2) xo + o → x + o2 [fast] (a) write the overall reaction. o3 + o → 2 o2 o3 + xo → x + 2 o2 xo + x + o3 + o → xo + x + 2 o2 x + o3 → xo + o2 (b) write the rate law for each step (using k for the rate constant). reaction 1: reaction 2: k[x][o2] k[xo][o2] k[x][o3] k[xo][o3] k[xo][o2] k[x][o] k[x][o2] k[xo][o] (c) x acts as a and xo acts as a catalyst intermediate intermediate catalyst (d) high-flying aircraft release no into the stratosphere, which catalyzes this process. when o3 and no concentrations are 3 ă— 1012 molecule/cm3 and 9.9 ă— 109 molecule/cm3, respectively, what is the rate of o3 depletion? the rate constant k for the rate-determining step is 6 ă— 10â’15 (cm3)2/moleculeâ·s. give your answer in scientific notation. use one significant figure in your answer. ă— 10 molecule/s
Answers: 2
You know the right answer?
1a. calculate the relative formula mass of magnesium oxide when the relative atomic masses are O=16...
Questions in other subjects:


Mathematics, 15.12.2019 01:31



Geography, 15.12.2019 01:31

Mathematics, 15.12.2019 01:31

Biology, 15.12.2019 01:31

Mathematics, 15.12.2019 01:31

Physics, 15.12.2019 01:31