

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:00, Unknowndragon42
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2

Chemistry, 22.06.2019 16:50, shaylawaldo11
Which element is least likely to undergo a chemical reaction
Answers: 3

You know the right answer?
For a gas sample containing equimolar amounts of carbon monoxideand heliumat 300 K, heliumhas averag...
Questions in other subjects:


Geography, 11.02.2021 03:00

Biology, 11.02.2021 03:00


Mathematics, 11.02.2021 03:00

Mathematics, 11.02.2021 03:00

Biology, 11.02.2021 03:00

Mathematics, 11.02.2021 03:00

Mathematics, 11.02.2021 03:00

Arts, 11.02.2021 03:00

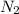 and
and 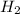 )
)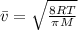
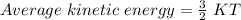 (not depend on molar mass)
(not depend on molar mass)

