
Chemistry, 15.07.2021 14:50 triciajfive
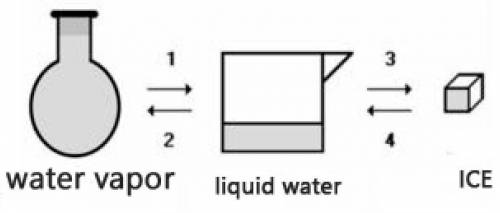
Consider the transformations a water sample undergoes without external pressure variation
(a) Transformations 2 and 4 are endothermic.
(b) Transformations 1 and 2 are exothermic.
(c) The amount of energy absorbed in 3 is equal to the amount released in 1.
(d) The amount of energy released in 2 is equal to the amount released in 4.
(e) The change of physical state does not involve heat energy.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:00, latoyatuggle23
Endeleev saw trends in the physical and chemical properties of elements when he organized them by
Answers: 2


Chemistry, 22.06.2019 22:30, teagan56
Gusing the milligrams of ascorbic acid you entered above, the ratio of total sample volume to aliquot volume, and the total milligrams of the vitamin c tablet that you dissolved, calculate the mass of ascorbic acid in the vitamin c tablet for each trial. do this by scaling up to find the amount (mg) of ascorbic acid in your 250 ml flask. enter your calculated mass of ascorbic acid in the vitamin c tablet, for each trial. be sure to enter your calculated mass in the corresponding order that you entered your milligrams of ascorbic acid. the milligrams of ascorbic acid you entered for entry #1 previously should correspond to the mass of ascorbic acid that you enter for entry #1 here.
Answers: 1

Chemistry, 23.06.2019 20:50, lindsay1054
Explain what happens to a gas when it is put into a container?
Answers: 2
You know the right answer?
Consider the transformations a water sample undergoes without external pressure variation
(a) Trans...
Questions in other subjects:



Mathematics, 03.12.2020 03:40




History, 03.12.2020 03:40


Business, 03.12.2020 03:40



