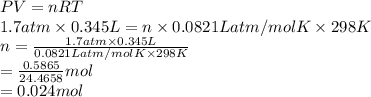Chemistry, 14.07.2021 01:00 hcarcel8287
An ideal gas has a temperature of 25 oC, a pressure of 1.7 atm, and volume of 345 mL, how many moles of gas are present in the container?

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 02:10, vapelordcarl69
When 225mg of anthracene, c14h10(s), was burned in a bomb calorimeter the temperature rose by 1.75k. calculate the calorimeter constant. by how much will the temperature rise when 125mg of phenol, c6h5oh(s), is burned in the calorimeter under the same conditions? (δch< (c14h10,s)=–7061 kj mol−1.)
Answers: 3

You know the right answer?
An ideal gas has a temperature of 25 oC, a pressure of 1.7 atm, and volume of 345 mL, how many moles...
Questions in other subjects:




Business, 21.06.2019 20:00

Mathematics, 21.06.2019 20:00

Mathematics, 21.06.2019 20:00




Mathematics, 21.06.2019 20:00

 = (25 + 273) K = 298 K
= (25 + 273) K = 298 K