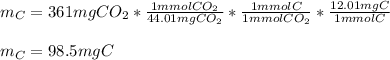Chemistry, 10.07.2021 15:00 cassidy32504
a pure sample of a compound on combustion analysis gave 361 mg of CO2 and 147mg of H2O. if the weight of the sample is 202 mg calculate the weight of carbon in the sample

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:00, actheorian8142
Flourine is found to undergo 10% radioactivity decay in 366 minutes determine its halflife
Answers: 3

Chemistry, 22.06.2019 08:30, dyanaycooper13
Analyze how limestone is weathered and identify the features that are formed as a result of this dissolution
Answers: 1


Chemistry, 22.06.2019 13:50, awesomegamergurl13
What happens when an atom of sulfur combines with two atoms of chlorine to produce sci2? a. each chlorine atom shares a pair of electrons with the sulfur atom. b. an electron is transferred from each chlorine atom to the sulfur atom. c. an electron is transferred from the sulfur atom to each chlorine atom. d. each chlorine atom shares all its valence electrons with the sulfur atom.
Answers: 2
You know the right answer?
a pure sample of a compound on combustion analysis gave 361 mg of CO2 and 147mg of H2O. if the weigh...
Questions in other subjects:



Mathematics, 03.12.2020 01:00


History, 03.12.2020 01:00

Mathematics, 03.12.2020 01:00

History, 03.12.2020 01:00

History, 03.12.2020 01:00