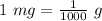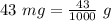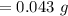43 mg = [?]g
A. 0.043 g
B. 4.3 g
C. 4300 g
D. 43,000 g
...

Chemistry, 09.07.2021 09:50 evanwall91
43 mg = [?]g
A. 0.043 g
B. 4.3 g
C. 4300 g
D. 43,000 g
![43 mg = [?]g A. 0.043 g B. 4.3 g C. 4300 g D. 43,000 g](/tpl/images/2421/0697/3cdd1.jpg)

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 08:30, vanessadaniellet21
Since the gas in your graduated cylinder is a mixture of butane and water vapor, you must determine the partial pressure of the butane, pbutane, alone. to do this, consult a reference and record the partial pressure of the water vapor, pwater, at the temperature you recorded. use the following formula to compute the partial pressure of the butane. pbutane = atmosphere - pwater use the following combined gas law formula and compute the volume that the butane sample will occupy at stp. (hint: convert both temperatures to kelvin.) pbutane x voriginal = pstandard x vfinal troom tstandard use the following ratio and proportion formula to determine the mass of butane needed to occupy a volume of 22.4 l at stp. grams of butane you used “x” grams of butane ml of butane corrected to stp = 22,400 ml compute the theoretical molar mass of butane based on its formula and the atomic masses on the periodic table. compare your experimental results from #3 to the theoretical value of #4, computing a percent error of your findings using this formula: % error = measured value - accepted value x 100 accepted value use the following ratio and proportion formula to determine the mass of butane needed to occupy a volume of 22.4 l at stp. need asap
Answers: 1

Chemistry, 23.06.2019 05:00, MoneyMike42
Select the statement that describe chemical properties a. antacid tablets neutralize stomach acid b. helium is the lightest monatomic element c. water freezes at 0 celsius d. mercury is liquid at room temperature
Answers: 3

Chemistry, 23.06.2019 13:30, gavianacandelar8522
The process by which liquid water changes into water vapour is called what ?
Answers: 2
You know the right answer?
Questions in other subjects:


Geography, 16.09.2019 10:50



Chemistry, 16.09.2019 10:50

Mathematics, 16.09.2019 10:50

Biology, 16.09.2019 10:50

History, 16.09.2019 10:50