
Chemistry, 06.07.2021 16:40 jenhorton2011
Using the image to the right how many oxygens will be present for water hint:chemical formula is H2O
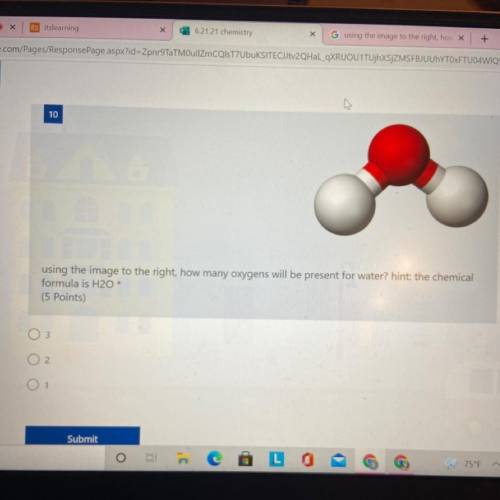

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:30, KieraKimball
10-14. (a) when 100.0 ml of weak acid ha were titrated with 0.093 81 m naoh, 27.63 ml were required to reach the equivalence point. find the molarity of ha. (b) what is the formal concentration of a- at the equivalence point? (c) the ph at the equivalence point was 10.99. find pk. for ha. (d) what was the ph when only 19.47 ml of naoh had been added?
Answers: 1

Chemistry, 22.06.2019 01:00, chrisxxxrv24
What are the variables in gay-lussac’s law? pressure and volume pressure, temperature, and volume pressure and temperature volume, temperature, and moles of gas
Answers: 1


Chemistry, 22.06.2019 22:00, genyjoannerubiera
What mass of glucose is produced when 54g of water react with carbon dioxide
Answers: 1
You know the right answer?
Using the image to the right how many oxygens will be present for water hint:chemical formula is H2O...
Questions in other subjects:




English, 08.04.2021 23:50


Mathematics, 08.04.2021 23:50


Biology, 08.04.2021 23:50

Mathematics, 08.04.2021 23:50



