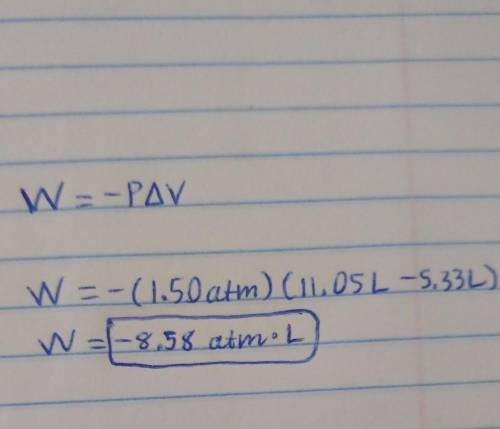Chemistry, 03.07.2021 01:20 wi8wuwj283jendjdudjd
A sample of gas is placed into an enclosed cylinder and fitted with a movable piston. Calculate the work (in joules) done by the gas if it expands from 5.33 L to 11.05 L against a pressure of 1.50 atm.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, elizediax8683
(apex) when a cup of water is dropped, as the cup falls, the water in the cup falls out true or false?
Answers: 1


Chemistry, 22.06.2019 08:40, jaueuxsn
Ageologist determines that a sample of a mineral can't be scratched by a steel nail but can be scratched by a masonry drill bit. based on this information, the sample mineral has to be softer than a. orthoclase. b. fluorite. c. apatite. d. corundum.
Answers: 2

Chemistry, 22.06.2019 20:30, ashley4329
Select all the correct answers. which compounds have the empirical formula ch20? (multiple answers)a. c2h4o2b. c3h603c. ch2o2d. c5h1005e. c6h1206
Answers: 2
You know the right answer?
A sample of gas is placed into an enclosed cylinder and fitted with a movable piston. Calculate the...
Questions in other subjects:




Biology, 06.07.2019 10:00

Social Studies, 06.07.2019 10:00





Social Studies, 06.07.2019 10:00