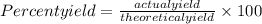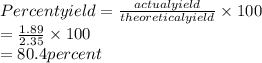Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:10, cameronbeaugh
13. a covalent bond between two atoms is likely to be polar if: a. one of the atoms is much more electronegative than the other. b. the two atoms are equally electronegative. c. the two atoms are of the same element. d. the bond is part of a tetrahedrally shaped molecule. e. one atom is an anion.
Answers: 1

Chemistry, 22.06.2019 14:30, hjlhdjfhjh
How does a noncompetitive inhibitor reduce an enzyme’s activity?
Answers: 1

Chemistry, 22.06.2019 15:00, tcapele252
‘which reaction would most likely require the use of an inert electrode?
Answers: 1
You know the right answer?
A student dissolved 3.50g of copper (II) nitrate in water and mixed it with a solution of sodium car...
Questions in other subjects:


Social Studies, 09.04.2020 04:35

Mathematics, 09.04.2020 04:35

History, 09.04.2020 04:35




Mathematics, 09.04.2020 04:36