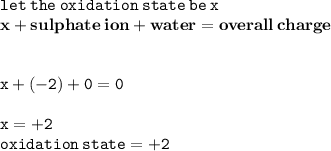Chemistry, 27.06.2021 16:30 rbrummitt8029
Calculate the oxidation number of the central atom of FeSO4 7H2O

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 23:10, carmenguabaoql9kv
Afusion reaction takes place between carbon and another element. neutrons are released, and a different element is formed. the different element is a) lighter than helium. b)heavier than helium. c)the same weight as helium. d)dependent on the element that reacted with carbon.
Answers: 3

Chemistry, 23.06.2019 06:40, hackman1216
The combustion of methane, ch4, releases 890.4kj/mol. that is, when one mole of methane is burned,890.4 kj are given off to the surroundings. this meansthat the products have 890.4 kj less than the reactants. thus, ah for the reaction = - 890.4 kj. a negative symbolforah indicates an exothermic reaction. ch (g) + 20 (g)> co2 (g) + 2 h0 (1); ah = - 890.4 kga) how much energy is given off when 2.00 mol of ch, are burned? b) how much energy is released when 22.4g of ch. areburned?
Answers: 1

You know the right answer?
Calculate the oxidation number of the central atom of FeSO4 7H2O...
Questions in other subjects:

Mathematics, 08.06.2020 04:57


English, 08.06.2020 04:57


English, 08.06.2020 04:57

Mathematics, 08.06.2020 04:57

English, 08.06.2020 04:57


History, 08.06.2020 04:57

History, 08.06.2020 04:57