
A student wants to make a 0.600 M aqueous solution of barium sulfate, BaSO4, and has a bottle containing 12.00 g of barium sulfate. What should be the final volume of the solution?
Find the numerical answer for this question and make sure to include the following:
What is the formula for molarity?
What is the molar mass for barium sulfate?
When you give your numerical answer, what are the correct significant figures and how do you know that is the correct amount?
Need this ASAP

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:00, porkhappycom
This line graph compares the growth of plants that were kept in the sun for different amounts of time.a) on day 7, the plants kept in the sun for 3 hours were how tall? b) on day 7, the plants kept in the sun for 6 hours were how tall? c) on day 10, the plants kept in the sun for 9 hours were how tall? d) on day 11, the plant that was grown with 1 hour of sunlight was how tall? e) based on the graph, the plant grows best in what amount of sunlight?
Answers: 1


Chemistry, 22.06.2019 09:10, cheesedoodle
When a nucleus absorbs a neutron and then breaks apart, there are many products of the reaction. what is not a product of a nuclear fission reaction
Answers: 1

Chemistry, 22.06.2019 10:00, alexabdercmur
Why is the structure of molecule important to its function?
Answers: 1
You know the right answer?
A student wants to make a 0.600 M aqueous solution of barium sulfate, BaSO4, and has a bottle contai...
Questions in other subjects:

Mathematics, 23.07.2019 02:00




Mathematics, 23.07.2019 02:00




Mathematics, 23.07.2019 02:00

Business, 23.07.2019 02:00

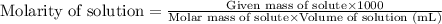 .....(1)
.....(1)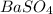 = 12.00 g
= 12.00 g![BaSO_4=[(1\times 137.33)+(1\times 32.07)+(4\times 16)]=233.4g/mol](/tpl/images/1384/4266/b0dcd.png)



