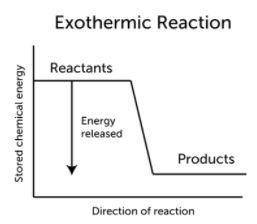
Which of the following is true of a reaction with a negative enthalpy?
O A. The enthalpy of the reactants is greater than that of the products.
B. The reaction has a negative activation energy.
C. The enthalpies of the products and reactants are both negative.
D. The enthalpy of the reactants is less than that of the products.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:00, mykalwashington
The compound methyl butanoate smells like apples. its percent composition is 58.8% c, 9.9% h, and 31.4% o. what’s the empirical formula ?
Answers: 1

Chemistry, 22.06.2019 03:00, Dkhaurithompson
Zoe is investigating the composition of substance a, an unknown substance. using chemical processes, she analyzes substance a and determines it is composed of sodium, oxygen, and hydrogen atoms in a ratio of 1 : 1 : 1. what is substance a? a. a compound b. an element c. a heterogeneous mixture d. a homogeneous mixture
Answers: 1

Chemistry, 22.06.2019 04:00, miamassimino
The rules of engagement (roe) working group is often used to (select all that apply.)
Answers: 2

Chemistry, 22.06.2019 21:30, crystalbyrd79p8imrx
What is the effect of returning nuclear reactor cooling water back into bodies of water?
Answers: 3
You know the right answer?
Which of the following is true of a reaction with a negative enthalpy?
O A. The enthalpy of the rea...
Questions in other subjects:


Mathematics, 09.04.2020 19:48


English, 09.04.2020 19:48

Mathematics, 09.04.2020 19:48

Mathematics, 09.04.2020 19:48


Chemistry, 09.04.2020 19:48

Mathematics, 09.04.2020 19:48