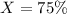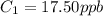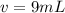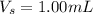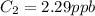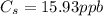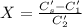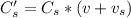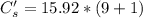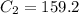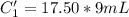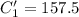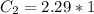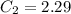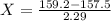A sample of drinking water was tested for Pb2 and was found to have a Pb2 concentration of 17.50 ppb. A 9.00 mL sample of the drinking water was spiked with 1.00 mL of a 2.29 ppb Pb2 standard. Analysis of the spiked sample gave a concentration of 15.93 ppb Pb2 . Find the percent recovery of the spike.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:00, dpazmembreno
Which describes interactions between substances and stomata during photosynthesis? check all that apply. oxygen enters stomata. oxygen is released through stomata. carbon dioxide enters stomata. carbon dioxide is released through stomata. hydrogen enters stomata. hydrogen is released through stomata.
Answers: 1

Chemistry, 22.06.2019 08:30, waterborn7152
Which common material is an example of a polymer? (25 pts) a. steel b. plastic c. petroleum d. rubbing alcohol
Answers: 2

Chemistry, 22.06.2019 22:10, steven0448
Determine the ph of 0.10 m nh3 solution. nh3 is a weak base with a kb equal to 1.8 x 10-5 round answer to nearest whole number.
Answers: 1
You know the right answer?
A sample of drinking water was tested for Pb2 and was found to have a Pb2 concentration of 17.50 ppb...
Questions in other subjects:


Law, 08.03.2021 19:40








Mathematics, 08.03.2021 19:40