
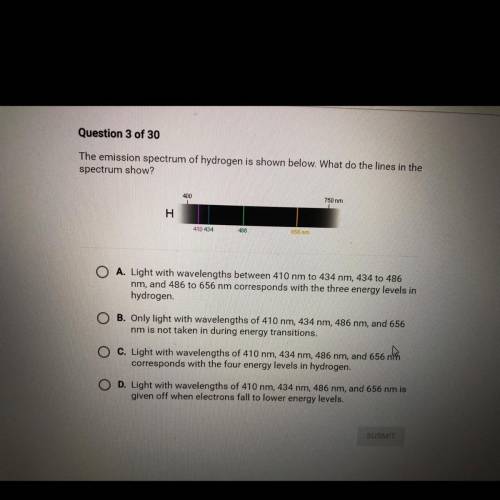
The emission spectrum of hydrogen is shown below. What do the lines in the
spectrum show?
A. Light with wavelengths between 410 nm to 434 nm, 434 to 486 nm, and 486 to 656 nm corresponds with the three energy levels in
hydrogen
B. Only light with wavelengths of 410 nm, 434 nm, 486 nm, and 656 nm is not taken in during energy transitions.
C. Light with wavelengths of 410 nm, 434 nm, 486 nm, and 656 no corresponds with the four energy levels in hydrogen.
D. Light with wavelengths of 410 nm, 434 nm, 486 nm, and 656 nm is given off when electrons fall to lower energy levels.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:00, orlando19882000
Ahypothrticalax type of ceramic material is known to have a density of 2.10 g/cm3 and a unit cell of cubic symmetry with a cell edge length of 0.57 nm. the atomic weights of the a and x elements are 28.5and 30.0 g/mol, respectively. on the basis of this information, which of the following crystal structures is (are) possible for this material: sodium chloride, cesium chloride, or zinc blende
Answers: 1

Chemistry, 22.06.2019 00:30, BLASIANNkidd
Maria wants to determine which type of disinfectant kills the most bacteria. which of the following is the best way for maria to determine this? a. ask ten different companies that make disinfectants which type is best. b. put the same amount and species of bacteria on ten identical plates, and add ten different kinds of disinfectant to each plate. c. interview ten different people to determine which type of disinfectant they prefer. d. put the same amount and species of bacteria on ten identical plates, and add a different disinfectant to each plate.
Answers: 1

Chemistry, 22.06.2019 14:00, njones58emailtjcedu
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 1

Chemistry, 22.06.2019 21:00, lalaween098
What type of radiation is lead emitting in the following equation? alpha particles beta particles gamma rays
Answers: 3
You know the right answer?
The emission spectrum of hydrogen is shown below. What do the lines in the
spectrum show?
Questions in other subjects:

Mathematics, 14.01.2022 06:30




Mathematics, 14.01.2022 06:30

World Languages, 14.01.2022 06:30






