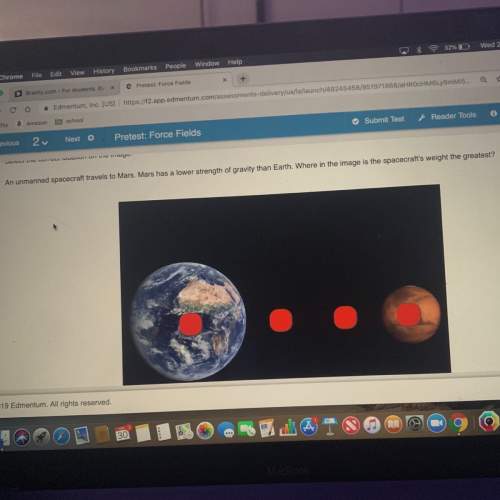
Chemistry, 23.06.2021 05:30 Zshotgun33
Given the balanced reaction: Zn + 2HCl → H2 + ZnCl2
If 5 grams of each reactant are available for the reaction and HCl is known
to be the limiting reactant, which of the following is correct?
O Both reactants will be completely used up.
O There will be excess of both reactants remaining.
O HCl will be completely used up while Zn will remain in excess.
O Zn will be completely used up while HCl will remain in excess.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:00, actheorian8142
Flourine is found to undergo 10% radioactivity decay in 366 minutes determine its halflife
Answers: 3

Chemistry, 22.06.2019 09:00, alydiale584
Particles vibrate in a rigid structure and do not move relative to their neighbors.
Answers: 1


Chemistry, 22.06.2019 18:00, meowmeowcow
Find the mass, in grams, of 5.00*10^23 molecules of f2
Answers: 3
You know the right answer?
Given the balanced reaction: Zn + 2HCl → H2 + ZnCl2
If 5 grams of each reactant are available for t...
Questions in other subjects:

Health, 03.02.2020 10:58



Mathematics, 03.02.2020 10:58

English, 03.02.2020 10:58


Chemistry, 03.02.2020 10:58


Social Studies, 03.02.2020 10:58