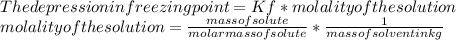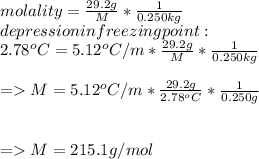Chemistry, 23.06.2021 02:50 jeffmacdonald1976
Suppose that you add 29.2 g of an unknown molecular compound to 0.250 kg of benzene, which has a K f of 5.12 oC/m. With the added solute, you find that there is a freezing point depression of 2.78 oC compared to pure benzene. What is the molar mass (in g/mol) of the unknown compound

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, catdog5225
Drive down any three characteristic of modern periodic table
Answers: 1



You know the right answer?
Suppose that you add 29.2 g of an unknown molecular compound to 0.250 kg of benzene, which has a K f...
Questions in other subjects:

Mathematics, 19.11.2020 02:10


Mathematics, 19.11.2020 02:10



Mathematics, 19.11.2020 02:10


Mathematics, 19.11.2020 02:10

Mathematics, 19.11.2020 02:10