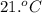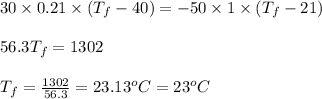Chemistry, 16.06.2021 08:00 Shadow0202
A 30. g sample of Aluminum was heated to 40. 0C and placed in a calorimeter containing 50. g of water at 21 0C. What is the final temperature of the aluminum-water system if the cAl = 0.21 cal/g0C and cwater = 1.0 cal/ g 0C.
Write the complete equation you will use. 1 point
Substitute the values in the equation in step 1 . 1 point
Report the math answer with 2 sig figs and the correct unit. 1 point

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 15:30, 20cschultz
Which suspect most likely committed the robbery and how do you know
Answers: 2

Chemistry, 22.06.2019 21:30, Turtlelover05
How can the periodic table be used to predict the behavior of elements?
Answers: 1
You know the right answer?
A 30. g sample of Aluminum was heated to 40. 0C and placed in a calorimeter containing 50. g of wate...
Questions in other subjects:

Mathematics, 07.03.2021 22:40

Mathematics, 07.03.2021 22:40

Mathematics, 07.03.2021 22:40




English, 07.03.2021 22:50

Social Studies, 07.03.2021 22:50

Mathematics, 07.03.2021 22:50

Mathematics, 07.03.2021 22:50

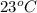
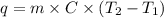
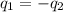
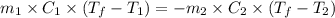 ......(1)
......(1)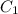 = specific heat of aluminium =
= specific heat of aluminium = 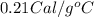
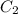 = heat capacity of water =
= heat capacity of water = 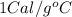
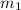 = mass of aluminium = 30. g
= mass of aluminium = 30. g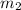 = mass of water = 50. g
= mass of water = 50. g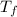 = final temperature of the system = ?
= final temperature of the system = ?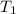 = initial temperature of aluminium =
= initial temperature of aluminium = 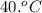
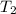 = initial temperature of the water =
= initial temperature of the water =