
Chemistry, 16.06.2021 07:30 babygirl123468
An inflated balloon contains X air molecules. After some time the volume of the balloon is found to be the half at the same temperature and pressure when a few air molecules are expelled out. a)How many molecules will be there in the balloon now? b) Which is the gas law associated with this?

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 21.06.2019 23:00, ayoismeisalex
Matches the chemical name of each oxide of phosphorus to its chemical formula
Answers: 2

Chemistry, 22.06.2019 02:30, BornAdopted21
Which piece of equipment would me most useful for measuring the volume of some water? a. pan balance b. graduated cylinder c. tweezers d. flask quick
Answers: 2
You know the right answer?
An inflated balloon contains X air molecules. After some time the volume of the balloon is found to...
Questions in other subjects:

Mathematics, 09.01.2021 02:30



Chemistry, 09.01.2021 02:40




Biology, 09.01.2021 02:40

English, 09.01.2021 02:40

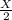 number of molecules now.
number of molecules now.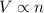 (At constant pressure and temperature)
(At constant pressure and temperature)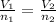 .....(1)
.....(1)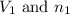 = Initial volume and number of moles
= Initial volume and number of moles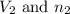 = Final volume and number of moles
= Final volume and number of moles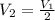
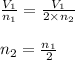
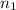 number of moles of gas contains X number of molecules
number of moles of gas contains X number of molecules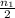 number of moles of gas will contain =
number of moles of gas will contain = 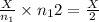 number of molecules
number of molecules

