
Chemistry, 15.06.2021 19:00 lilpeepxliltracy
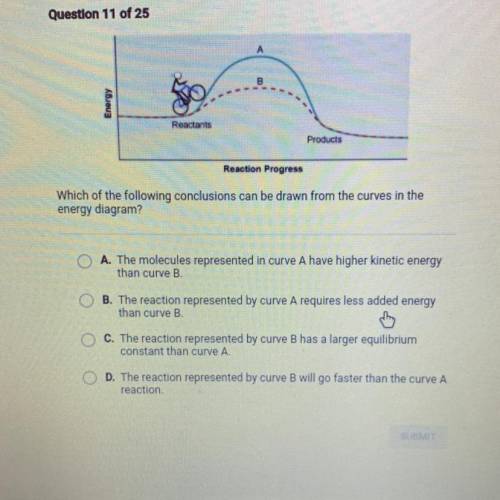
Question 11 of 25
Reactants
Products
Reaction Progress
Which of the following conclusions can be drawn from the curves in the
energy diagram?
O A. The molecules represented in curve A have higher kinetic energy
than curve B.
OB. The reaction represented by curve A requires less added energy
than curve B.
dh
C. The reaction represented by curve B has a larger equilibrium
constant than curve A.
D. The reaction represented by curve B will go faster than the curve A
reaction

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 20:00, aksambo4707
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1

Chemistry, 22.06.2019 20:00, bettybales1986
Glucose (c6h12o6) is an important biological molecule. (round the answer to nearest hundredth.) what is the percent by mass of carbon in glucose?
Answers: 2

Chemistry, 22.06.2019 22:30, wpatskiteh7203
Draw the aromatic compound toluene (methylbenzene). show all hydrogen atoms, including those on the ring.
Answers: 1
You know the right answer?
Question 11 of 25
Reactants
Products
Reaction Progress
Which of the following con...
Products
Reaction Progress
Which of the following con...
Questions in other subjects:




Physics, 17.06.2020 21:57

Mathematics, 17.06.2020 21:57

Health, 17.06.2020 21:57

Mathematics, 17.06.2020 21:57



English, 17.06.2020 21:57



