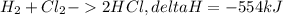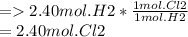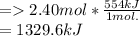Chemistry, 14.06.2021 15:50 yohannaG4315
How much energy would be produced from the reaction of 2.40 moles hydrogen with 3.95 moles chlorine? ___ H2 + ___ Cl2 ___ HCl ΔH = -554 kJ

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:30, jameskarbar9p8c9d2
Match the following vocabulary terms to their definitions. 1. amount of energy required to change 1 gram of material from the solid to the liquid state at its melting point 2. a measure of the kinetic energy of the particles of a substance 3. the amount of heat energy required to raise the temperature of 1 gram of liquid water from 14.5°c to 15.5°c 4. amount of energy required to change 1 gram of material from the liquid to the gaseous state at its boiling point 5. the amount of energy required to change 1 gram of a substance 1°c a. temperature b. latent heat of vaporization c. latent heat of fusion d. calorie e. specific heat
Answers: 1

Chemistry, 22.06.2019 16:30, jrfranckowiak
At 20°c, a sample of h2o liquid and a sample of co2 gas each have the same average kinetic energy. why is one a liquid and the other a gas at this temperature?
Answers: 1

Chemistry, 22.06.2019 17:20, banna01man
Pegmatites are igneous rocks in which the individual minerals are very large. typically, the minerals are all light-colored quartz, feldspar and muscovite. if you were given a black and white photograph of a pegmatite in a quarry (where the rock has been blasted and broken), what physical properties could you use to identify those three minerals in this hypothetical photo? describe each mineral and the specific diagnostic properties. be specific.
Answers: 2
You know the right answer?
How much energy would be produced from the reaction of 2.40 moles hydrogen with 3.95 moles chlorine?...
Questions in other subjects:



Mathematics, 22.05.2020 01:01


Social Studies, 22.05.2020 01:01

Spanish, 22.05.2020 01:01


Mathematics, 22.05.2020 01:01

English, 22.05.2020 01:01