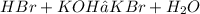Chemistry, 10.06.2021 23:10 jocelynfray16
Assume that the reaction of aqueous hydrobromic acid solution and potassium hydroxide base undergoes a complete neutralization reaction.
a. Write a balanced chemical equation.
b. How many grams of water can be produce from the complete reaction of excess hydrobromic acid and 55.4 mL of 0.685 M potassium hydroxide solution, assuming that potassium hydroxide is the limiting reactant?
A solution of hydrobromic acid is formed by dissolving 5.00 grams in enough water to make 1.5 L solution.
c.. What was the molarity of this solution?
d. What is the pH of this solution?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:00, jasminortega2002
Which of the following compounds does not contain molecules? question 2 options: co2 h2 nacl h2o
Answers: 1


Chemistry, 22.06.2019 21:50, SoccerAllStar2
Liquid from a brewery fermentation contains 10% ethanol and 90% water. part of the fermentation product (50,000 kg/h) is pumped to a distillation column on the factory site. under current operating conditions, a distillate of 45% ethanol and 55% water is produced from the top of the column at a rate of one-tenth that of the feed. what is the composition of the waste "bottoms" from the still?
Answers: 2

Chemistry, 23.06.2019 00:30, natishtaylor1p8dirz
What is the chemical formula of magnesium bromide? a. mgbr2 b. mgbr c. mg2br2 d. mg2br
Answers: 3
You know the right answer?
Assume that the reaction of aqueous hydrobromic acid solution and potassium hydroxide base undergoes...
Questions in other subjects:

Social Studies, 20.09.2020 17:01

English, 20.09.2020 17:01



History, 20.09.2020 17:01

Mathematics, 20.09.2020 17:01

Mathematics, 20.09.2020 17:01


Mathematics, 20.09.2020 17:01