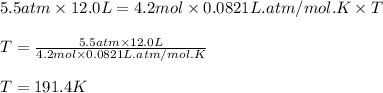Chemistry, 10.06.2021 09:10 ianbrown00121
(PLEASE HELP 20 POINTS)
4.2 moles of an unknown gas at a pressure of 5.5 atm has a volume of 12.0 L.
What is the temperature?

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:30, cheyennecarrillo14
If you add 5.00 ml of 0.100 m sodium hydroxide to 50.0 ml of acetate buffer that is 0.100 m in both acetic acid and sodium acetate, what is the ph of the resulting solution? acetic acid: ka = 1.8. x 10-5
Answers: 1


Chemistry, 23.06.2019 01:40, mandilynn22
Calcium carbonate decomposes at high temperatures to give calcium oxide and carbon dioxide as shown below. caco3(s) cao(s) + co2(g) the kp for this reaction is 1.16 at 800°c. a 5.00 l vessel containing 10.0 g of caco3(s) was evacuated to remove the air, sealed, and then heated to 800°c. ignoring the volume occupied by the solid, what will be the mass of the solid in the vessel once equilibrium is reached?
Answers: 1
You know the right answer?
(PLEASE HELP 20 POINTS)
4.2 moles of an unknown gas at a pressure of 5.5 atm has a volume of 12.0 L...
Questions in other subjects:





Mathematics, 28.02.2021 04:40


Mathematics, 28.02.2021 04:40

Mathematics, 28.02.2021 04:40

Biology, 28.02.2021 04:40

Mathematics, 28.02.2021 04:40

 .......(1)
.......(1)