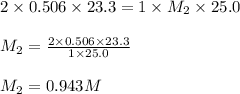Chemistry, 09.06.2021 23:20 AutumnJoy12
Calculate the molarity of a potassium hydroxide solution if 25.0 ml of this solution was completely neutralized by 23.3 ml of 0.506 M sulfuric acid.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:20, ayoismeisalex
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 16:30, Eddie997
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2

Chemistry, 22.06.2019 22:30, COOLIOMARIS
What three things does a balanced equation show you?
Answers: 1
You know the right answer?
Calculate the molarity of a potassium hydroxide solution if 25.0 ml of this solution was completely...
Questions in other subjects:










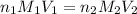 ........(1)
........(1)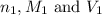 are the n-factor, molarity and volume of acid that is
are the n-factor, molarity and volume of acid that is 
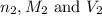 are the n-factor, molarity and volume of the base that is KOH
are the n-factor, molarity and volume of the base that is KOH