Chemistry, 09.06.2021 08:50 bbenaventbbbb9653
Enough nitrogen must be generated in the bag to create a total pressure of 2.56 atm, which then drops as the bag slowly deflates. Assuming the volume of the bag is 56.0 L and the temperature in the car is 25oC, calculate the molar quantity (number of moles) of N2 that must be generated. Hint: Use Ideal gas law PV=nRT (Ideal gas constant R=0.080 L. atm/mol. K)

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:00, tamikagoss22
Agas in a balloon at constant pressure has a volume of 160 ml at -125*c. what is its volume at 29.0*c?
Answers: 1

Chemistry, 23.06.2019 04:10, nabeelunique
An unknown substance has been shown to have metallic bonds. which of the following is most likely a property of this substance? a. low conductivity b. low boiling point c. high malleability d. high solubility in water
Answers: 2

Chemistry, 23.06.2019 06:00, bvbbridesmaid5519
Is the flow of energy during vaporizing more like the flow during melting or during freezing
Answers: 1
You know the right answer?
Enough nitrogen must be generated in the bag to create a total pressure of 2.56 atm, which then drop...
Questions in other subjects:

Social Studies, 26.11.2019 11:31

Biology, 26.11.2019 11:31

Mathematics, 26.11.2019 11:31



History, 26.11.2019 11:31

Business, 26.11.2019 11:31


English, 26.11.2019 12:31

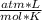 T= 25 C= 298 K (being 0 C= 273 K)
T= 25 C= 298 K (being 0 C= 273 K)