
Many computer chips are manufactured from silicon, which occurs in nature as SiO2. When SiO2 is heated to melting, it reacts with solid carbon to form liquid silicon and carbon monoxide gas. In an industrial preparation of silicon, 155.0 kg of SiO2 is allowed to react with 78.2 kg of carbon to produce 66.0 kg of silicon.
Required:
a. What is the theoretical yield for the reaction in Kg Si?
b. What is the percent yield for the reaction?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:40, bferguson7986
What kind of ion is contained in salts that produce an acidic solution? a positive ion that attracts a proton from water a positive ion that releases a proton to water a negative ion that attracts a proton from water a negative ion that releases a proton to water
Answers: 1

Chemistry, 22.06.2019 10:40, justicejesusfreak
Which buffer would be better able to hold a steady ph on the addition of strong acid, buffer 1 or buffer 2? explain. buffer 1: a solution containing 0.10 m nh4cl and 1 m nh3. buffer 2: a solution containing 1 m nh4cl and 0.10 m nh3
Answers: 1

Chemistry, 22.06.2019 12:00, winterblanco
What is the lowest number energy level where a d sublevel is found
Answers: 1
You know the right answer?
Many computer chips are manufactured from silicon, which occurs in nature as SiO2. When SiO2 is heat...
Questions in other subjects:

Health, 09.07.2019 11:00



English, 09.07.2019 11:00






English, 09.07.2019 11:00

 .....(1)
.....(1) :
: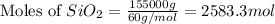
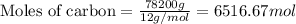

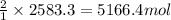 of carbon
of carbon of silicon
of silicon
 ......(2)
......(2)



