
Chemistry, 08.06.2021 16:40 AbigailHaylei
4Fe (s) + 3 O2 (g) → 2 Fe2O3 (s)
A. What is the oxidation number for each item in this equation?
B. What is being oxidized?
C. What is being reduced?
D. What is the oxidizing agent?
E. What is the reducing agent?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 13:00, yaneiryx5476
Is 9 correct? and can someone me with 10? it’s due tomorrow, you
Answers: 1

Chemistry, 22.06.2019 21:00, rhondafits9000
Which property of water causes water drops to bead on a freshly waxed car?
Answers: 2


Chemistry, 23.06.2019 11:40, markfaerman5408
An electron moved from a lower energy level to a higher energy level. what most likely happened during the transition? a random amount of light was released. a fixed amount of energy was absorbed. a fixed amount of energy was released. a random amount of light was absorbed.
Answers: 1
You know the right answer?
4Fe (s) + 3 O2 (g) → 2 Fe2O3 (s)
A. What is the oxidation number for each item in this equation?
Questions in other subjects:

Mathematics, 18.09.2020 03:01

Mathematics, 18.09.2020 03:01

Mathematics, 18.09.2020 03:01

Mathematics, 18.09.2020 03:01

Mathematics, 18.09.2020 03:01

Mathematics, 18.09.2020 03:01

Mathematics, 18.09.2020 03:01

Mathematics, 18.09.2020 03:01

Mathematics, 18.09.2020 03:01

English, 18.09.2020 03:01

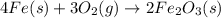
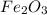 is +3.
is +3. 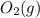 is 0 as it is present in its elemental state.
is 0 as it is present in its elemental state.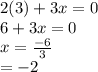
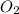 as a decrease in its oxidation state is occurring. So,
as a decrease in its oxidation state is occurring. So, 

