
Chemistry, 08.06.2021 03:20 avisconti571
A sample of 0.2140 g of an unkown substance monoprotic acid was dissolved in 25.0 mL of water and titrated with 0.950 M NaOH. The acid required 27.4 mL of base to reach the equivalence point. After 15.0 mL of base had been added in the titration, the pH was found to be 6.50. What is the Ka for the unknown acid?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:30, sbhishop19
Balance this equation co2(g) + h2o (g) show that the balanced equation obeys the law if conversation of mass
Answers: 1


Chemistry, 23.06.2019 09:30, tramqpham25
People who practice which of the following diets may run the risk of not getting enough iron. a. gluten free or vegan diet b. diet for managing diabetes c. vegan diet d. gluten free diet
Answers: 2

Chemistry, 23.06.2019 18:20, anaroles04
Asalt is best described as a compound that is formed from the reaction between an acid and a base. a strong acid and a weak acid. a strong base and a weak base. an acid and water save and exit next submit
Answers: 1
You know the right answer?
A sample of 0.2140 g of an unkown substance monoprotic acid was dissolved in 25.0 mL of water and ti...
Questions in other subjects:

Mathematics, 26.09.2019 19:10


Mathematics, 26.09.2019 19:10

Chemistry, 26.09.2019 19:10


Mathematics, 26.09.2019 19:10




Mathematics, 26.09.2019 19:10

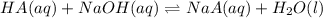
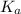 of HA
of HA
![K_a=\frac{[A^-].[H^+]}{[HA]}](/tpl/images/1366/2932/3c83d.png)
![$[HA] = \frac{^nH_A}{V}$](/tpl/images/1366/2932/b0ec6.png)
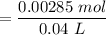
![$[NaOH]= \frac{0.015L \times 0.0950 M}{V}$](/tpl/images/1366/2932/a5013.png)
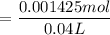

 and 0.0356 M
and 0.0356 M 
![[H^+]](/tpl/images/1366/2932/07acb.png)
![$[H^+] = 10^{-pH}$](/tpl/images/1366/2932/d6eff.png)


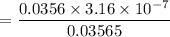

 .
.

