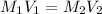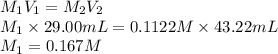Chemistry, 07.06.2021 15:30 biggiecheese93
A 29.00 mL sample of an H2SO4 solution of unknown concentration is titrated with a 0.1122 M KOH solution. A volume of 43.22 mL of KOH was required to reach the equivalence point. What is the concentration of the unknown H2SO4 solution? Express your answer using four significant figures.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, officialgraciela67
Embryos of different species look very similar, which shows that the organisms share a ancestor.
Answers: 1

Chemistry, 22.06.2019 19:30, amandamiro05
Helium decays to form lithium. which equation correctly describes this decay?
Answers: 2


Chemistry, 23.06.2019 02:00, bagofmud8339
The point along a planet's orbit where it is closest to the sun is called the
Answers: 1
You know the right answer?
A 29.00 mL sample of an H2SO4 solution of unknown concentration is titrated with a 0.1122 M KOH solu...
Questions in other subjects:


Health, 02.12.2020 23:00



Mathematics, 02.12.2020 23:00



Chemistry, 02.12.2020 23:00

Biology, 02.12.2020 23:00

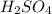 solution is 0.167 M.
solution is 0.167 M.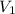 = 29.00 mL,
= 29.00 mL, 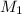 = ?
= ?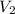 = 43.22 mL,
= 43.22 mL, 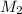 = 0.1122 M
= 0.1122 M