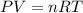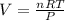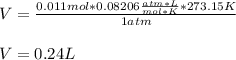Chemistry, 04.06.2021 01:10 deanlmartin
A chemical reaction done at STP conditions is supposed to produce 0.011 moles of CO2 gas. How many liters of gas will that be?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:30, dpchill5232
Suppose a lab group reports a ppercent yield of sand of 105. is it really possible to collect more sand than was originally represented? what is the possible explanation for the extra product?
Answers: 2

Chemistry, 22.06.2019 13:10, kellinvagneur
Which electron configuration represents the electrons in an atom of sodium in the ground state at stp
Answers: 1

You know the right answer?
A chemical reaction done at STP conditions is supposed to produce 0.011 moles of CO2 gas. How many l...
Questions in other subjects:



Biology, 28.08.2019 14:30

History, 28.08.2019 14:30


Mathematics, 28.08.2019 14:30

Biology, 28.08.2019 14:30